article- German Deaths Analysis 2022
Article: German Deaths Data Analysis 2022
Re: the alarming increase in deaths since the covid injection rollout in Germany & the failure of the PEI & RKI to do this analysis
Re-post of original article: (as reported on thegatewaypundit.com Dec 13, 2022)
German Data Analyst Reveals Data from Health Insurance Shows 4 Times Increase in Sudden Deaths Following COVID Vaccine Rollouts
by Jim Hoft
German data analyst Tom Lausen held a conference on Monday in the Bundestag to discuss the massive rise in people who died “suddenly and unexpectedly” after the Covid vaccine rollout.
Only one mainstream journalist was present during the press conference.
The National Association for Statutory Health Insurance Physicians (NASHIP) provided the data covering over 72 million insured Germans.
NASHIP or “Kassenärztliche Bundesvereinigung (KBV),” is the coordinating body of all 17 State Associations of Statutory Health Insurance Physicians in Germany. As of 2018, it represented about 175,000 office-based physicians and psychotherapists.
“The association is also actively involved in negotiations on the range of services provided by statutory health insurance companies and the remuneration of doctors. Furthermore, the KBV carries out a “security mandate” in accordance with Section 75 (1) SGB V, which is intended to ensure that all legally insured patients can receive adequate outpatient care.”
The government agency responsible with assessing the risks of vaccination apparently did not inquire about this information, as stated during the news conference.
“According to Section 13 of the Infection Protection Act, the Robert Koch Institute and the Paul Ehrlich Institute have to evaluate diagnosis codes from the National Association of Statutory Health Insurance Physicians (KBV) at reqular intervals in order to determine vaccination effects. But since the beginning of the corona vaccinations, this data has neither been requested nor evaluated.”
“After numerous inquiries in recent months, the KBV data (period: 2016 to the first quarter of 2022) are available to our health policy spokesman, Martin Sichert,” according to the website.
More from afdbundestag.de:
Since the beginning of the corona vaccination, there have been drastic changes in the number of diseases and deaths in the population. This is based on data from the Association of Statutory Health Insurance Physicians, which Martin Sichert was able to evaluate exclusively together with data expert Tom Lausen and which will be presented at the press conference.
Patient data from the National Association of Statutory Health Insurance Physicians (KBV) on the side effects of corona vaccinations provide frightening insights: With the start of mass corona vaccinations, the number of people who died “suddenly and unexpectedly” skyrocketed compared to previous years, more than fourfold. In every quarter, starting with the first quarter of 2021, more sudden and unexpected deaths were identified by panel doctors than in every year from 2016 to 2020 as a whole.
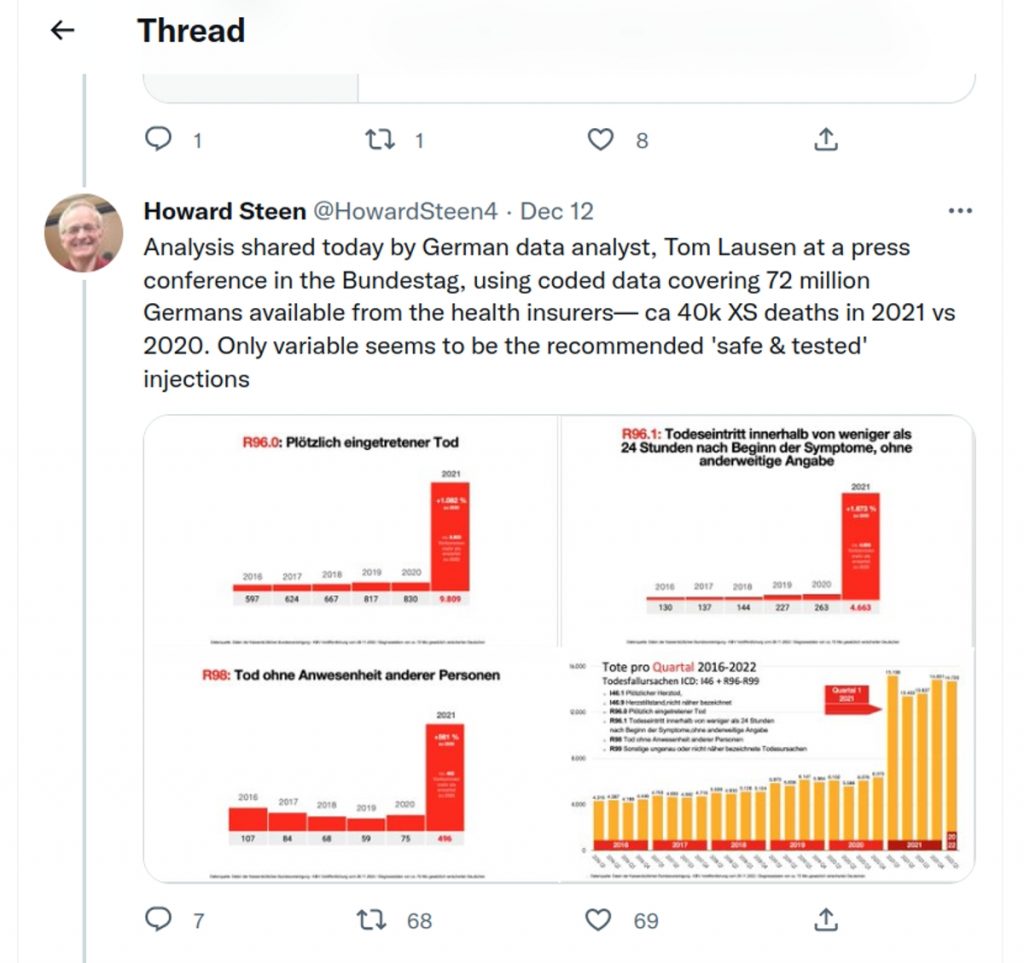
Using coded data covering 72 million Germans available from the health insurers, the number of people who died “suddenly and unexpectedly” skyrocketed compared to previous years.
“It was found that in 2021 not only were 2,487,526 patients with vaccination side effects seen by the doctor, but that there were also drastic changes in clinical pictures and deaths since the start of the corona vaccination.”
The following diagnostic keys were evaluated in order to analyze the rise in sudden deaths:
- R96.0 Sudden death
- R96.1 Death occurring within less than 24 hours of onset of symptoms, unless otherwise stated
- R98 Death without others present
- R99 Other imprecise or unspecified causes of death
- I46.1 Sudden cardiac death
Report24 reported:
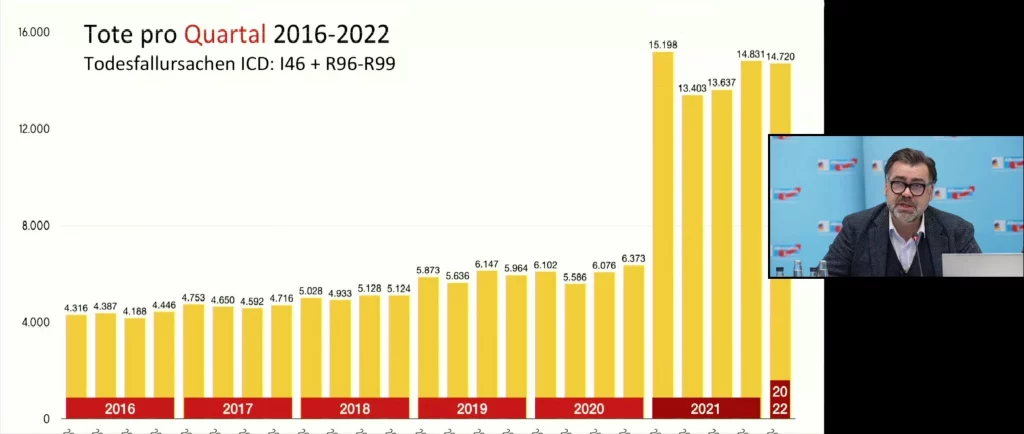
The following graphic illustrates the sharp increase in diagnoses R96 to R99 since the first quarter of 2021:
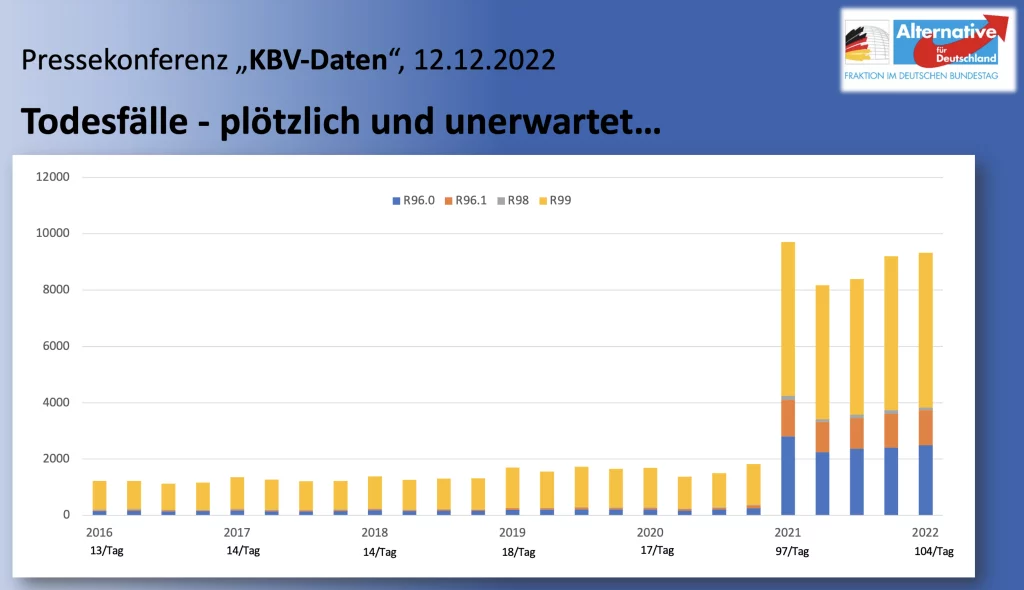
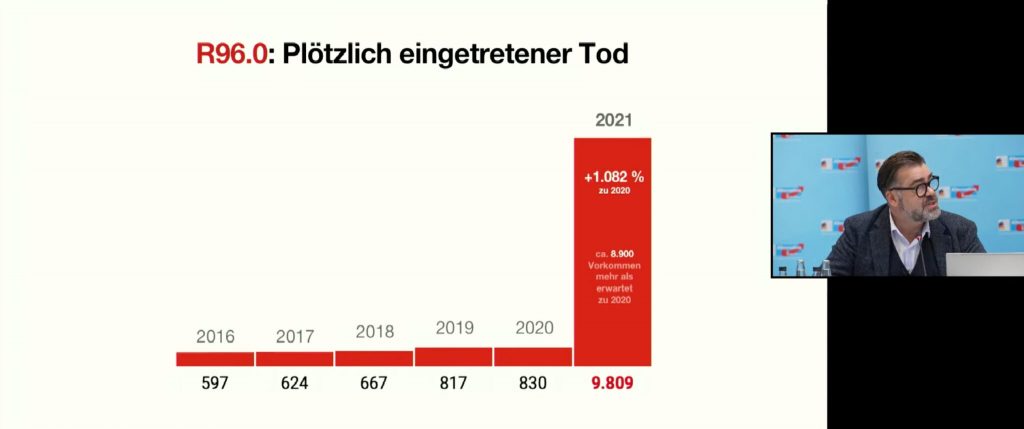
According to KBV data, in 2021 there was an increase in diagnoses of “sudden death” (R96) of +1,082 percent:
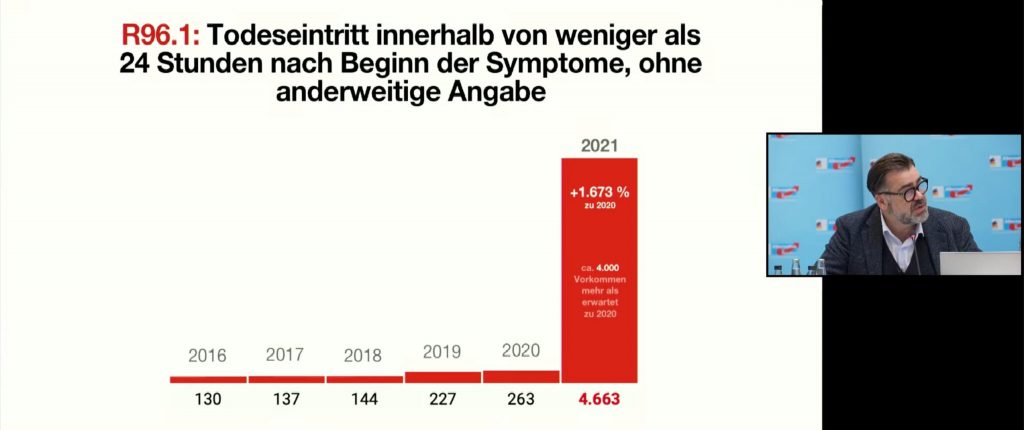
With diagnostic key R96.1, the increase is even +1,673 percent:
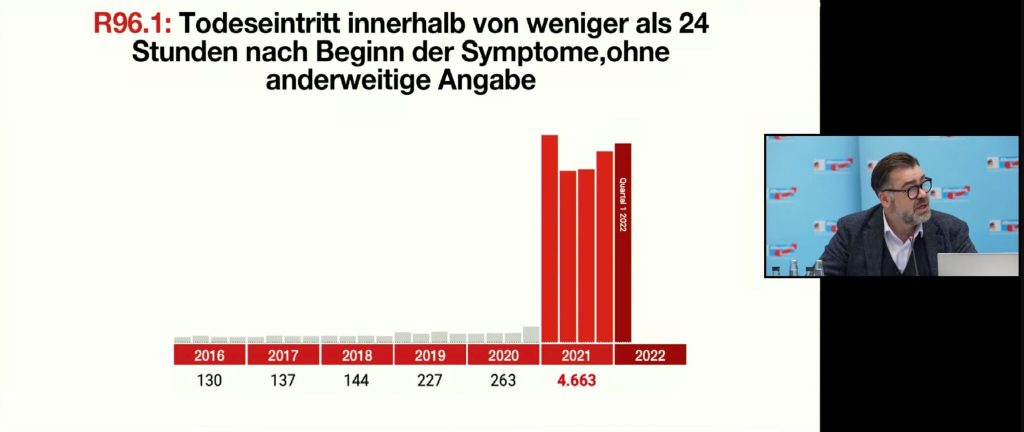
If you look at the figures per quarter, a first slight increase can already be seen at the end of 2020:
The diagnosis of “sudden cardiac death” has also been made significantly more frequently since 2021:
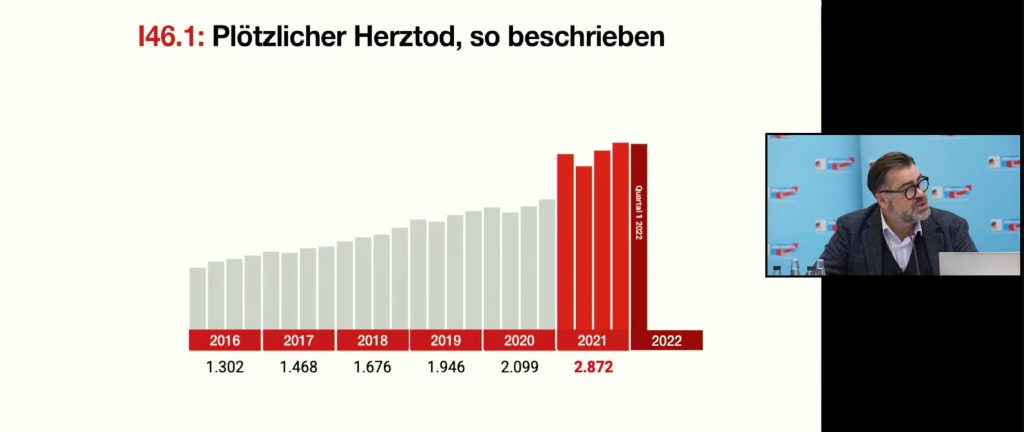
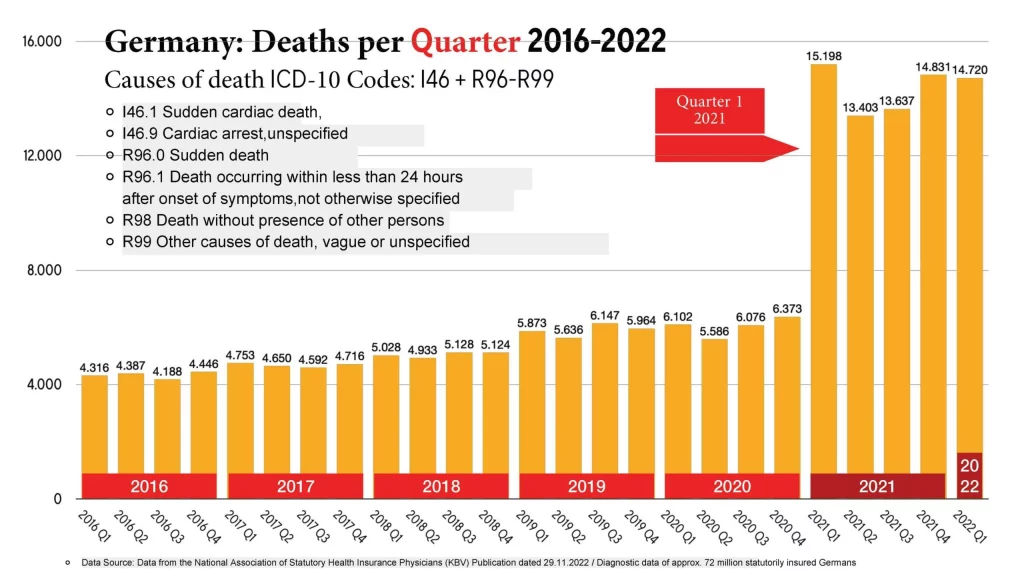
Lausen presented the following overview of the increase in deaths by quarter for ICD I46 and R96 to R99 at the press conference:
The team of analysts demanded that:
- Immediate suspension of vaccination with the corona vaccines until it can be ruled out that the massive increase in deaths is due to the vaccination
- Autopsies on all those who died suddenly and unexpectedly to determine where they came from massive increase coming
- Mandatory recording of the vaccination status of the corona vaccinations and the used vaccine in all deceased and regular publication of this data
- Immediate evaluation of the KB data by the PEI and RKI and information of the population and doctors about the increase in diseases
- Linking of the KBV data with the vaccination data by PEI and RKI and publication
The German PDF data is available to download here.
You can watch the conference below:
Related Videos
related articles
- see more in Changing Definitions: Vaccine Safety
- see the Grand Jury Day 4: the Injection
- Indiana life insurance CEO says deaths are up 40% among people ages 18-64
- German Insurance letter to PEI: Vaccine Injuries are under-reported
- see more Vaccine info in Links
Keywords
accounting, Afd, article, Autopsies, Data, deaths, evaluation, German Infection Protection Act, germany, Großenbach, Health Insurance, INEK, Infection Protection Act, Insurance, Kassenärztliche Bundesvereinigung, KBV, Lausen, myocarditis, NASHIP, National Association for Statutory Health Insurance Physicians, Paul Ehrlich Institute, PE, Plötzlich, press conference, risks, RKI, Robert Koch Institute, Safety, Sichert, side effects, Sudden death, underreporting, Unerwartet, Vaccination, Vaccine